
Government of the Virgin Islands - High Blood Pressure Treatment Drug Valsartan Recalled Healthcare professionals and patients are being advised of a voluntary recall of products containing the active pharmaceutical ingredient (API)

Teva Issues Voluntary Nationwide Recall of One Lot of Anagrelide Capsules, USP 0.5 mg Due to Dissolution Test Failure | FDA
Teva Issues Voluntary Nationwide Recall of One Lot of Anagrelide Capsules, USP 0.5 mg Due to Dissolution Test Failure
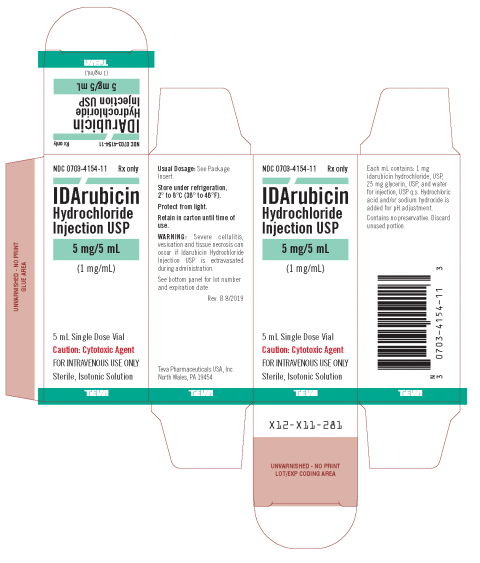
Teva Issues Voluntary Nationwide Recall of One Lot of IDArubicin Hydrochloride Injection USP 5 mg/5 mL Due to the Presence of Particulate Matter | FDA






/cloudfront-us-east-2.images.arcpublishing.com/reuters/CCBS56SBRRKUDM7JEVVE3UOQEE.jpg)




/cloudfront-us-east-1.images.arcpublishing.com/gray/UVAUY3N3VBKKPJZ24MSVDZL62I.png)




